Academic Research
Select the right study participants, quickly and effectively.
Getting your research project off on the right foot depends on including the right participants and excluding the wrong ones. Proem's licensed digital mental health screening and assessment tools help researchers increase the accuracy of participant identification. And results are scored within minutes, drastically decreasing the time needed for recruitment.
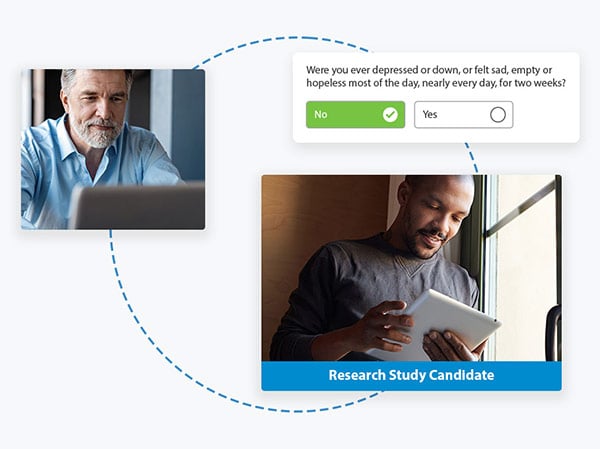
A few of our licensed digital tools include:
-
Mini International Neuropsychiatric Interview (M.I.N.I.)
-
M.I.N.I. Kid
-
Sheehan Disability Scale (SDS)
-
Yale-Brown Obsessive Compulsive Scale (YBOCS)
-
Body Dysmorphic Disorder Questionnaire (BBDQ)
-
And more...
Rely on the Most Trusted Scales in Research
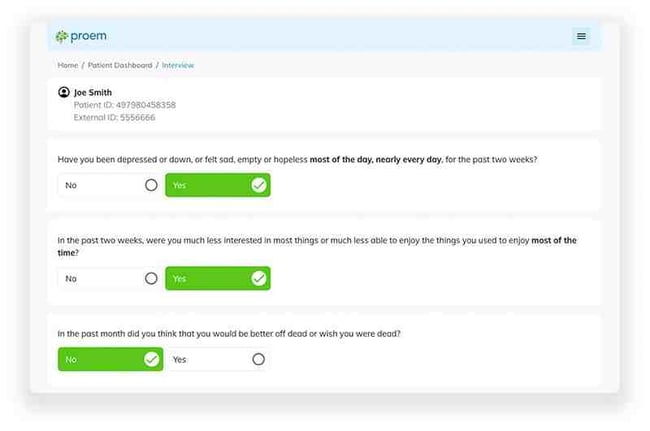
Gold-Standard Scales Help Accurately Identify Candidates
Proem is the exclusive digital licensee worldwide of clinically validated scales written by world-renowned psychiatrists to screen for the most common DSM-5 mental health disorders. The Proem licensed solution scales, including the Mini international Neuropsychiatric Interview (M.I.N.I.), SDS, S-STS, YBOCS I&II, CYBOCS I&II, BDDQ, and others, are used to accurately identify underlying conditions in study candidates that can materially impact study time and risk.

Thousands of Citations Provide Safety in Numbers
The tools that Proem has licensed have been cited in more than 175,000 articles and used in worldwide studies reviewed by the FDA, National Institutes of Health (NIH), National Institutes of Mental Health (NIMH), World Health Organization (WHO), European Pharmaceutical Associations, and Federation of Asian Pharmaceutical Associations.
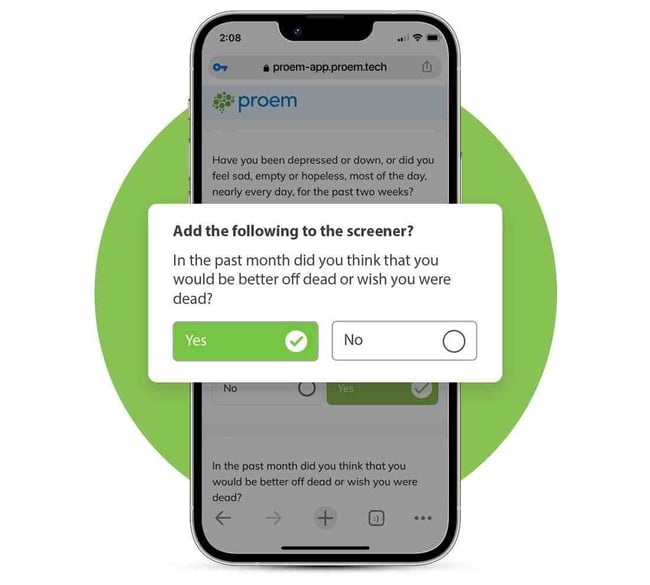
Customize the Proem Tool to Match Your Study Subject
The Proem solution is adaptable, enabling researchers to tailor the screeners and assessments to their studies. This makes it faster for participants to complete the assessments, thereby decreasing the time to identify candidates.
Solution
Proven Digital Tools for Research Study Participant Selection
Proem’s software platform features exclusively licensed digital tools that have been used by academic research organizations worldwide to increase the accuracy of participant identification and decrease the time needed for patient recruitment. Using digital mental health screeners and assessments for inclusion/exclusion criteria offers multiple benefits over paper-based tools.
Using one digital tool, Proem can evaluate the participant for the most common DSM-5 mental disorders at one time, eliminating the need to use multiple, time-consuming tools.
Proem's digital tools automatically dig deeper into specific disorders or skip a disorder altogether based on how the person answers each question. This capability helps gather more accurate screening data for more reliable results.
Proem’s digital tools can guide a researcher or participant through interview questions in a structured way that leaves no room for interpretation or manipulation. Removing rater bias can help decide whether to include or exclude a person from a research study.
Digital screeners and interviews can be sent to participants to complete electronically at their convenience rather than in person, improving patient compliance and saving time. Participants also may be more truthful when answering questions on their own.
Complex paper assessments are difficult to score and lend themselves to human error. Proem’s digital solution automatically scores assessments in a matter of seconds, mitigating scoring errors and helping ensure accurate results in near real-time.
Proem’s digital behavioral health screening and assessment solution can track participants during a study to automatically aggregate and measure outcomes to assist with publishing study findings.
Proem Behavioral Health is the exclusive licensee of Dr. David Sheehan's behavioral assessment tools in digital, electronic and pdf format and is authorized to sublicense that right to third parties. Proem Behavioral Health maintains concurrent rights to distribute or license paper forms of the Sheehan Technology.
Assessments
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
| Anxiety Disorder Screener | Determines if a patient may have an anxiety disorder, including Panic, Agorophobia, Social Anxiety, OCD, PTSD, GAD (9 questions) | Anxiety | Adult | Provider or Patient (typically) |
| BDDQ (Body Dysmorphic Disorder Questionnaire) | Determines if a person may have Body Dysmorphic Disorder | BDD | Adult | Patient |
| C-FOCI (Children's Obsessive Compulsive Inventory) | OCD symptoms experienced in the past month (24 questions) | OCD | Children & Adolescents | Provider or Patient |
| C-SSRS (Columbia Suicide Severity Rating Scale) | Suicidality screener (5 questions) | Suicidality | Adult | Provider or Patient (typically) |
| Eating Disorder Screener | Determines if a patient may have an eating disorder, including Anorexia, Bulimia, Binge Eating (4 questions) | Eating Disorders | Adult | Provider or Patient (typically) |
| FOCI (Florida Obsessive Compulsive Inventory) | OCD symptoms experienced in the past month (25 questions) | OCD | Adult | Provider or Patient |
| GAD-7 (Generalized Anxiety Disorder-7) | 7 questions used to measure the level of Generalized Anxiety Disorder symptoms | Anxiety | Adult | Provider or Patient (typically) |
| Kid Mood Disorder Screener Ages 13-17 | Determines if a patient may have a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders (9 questions) | Mood Disorders | Adolescents | Provider or Patient (typically) |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Adult Screener | Screens for 17 behavioral health categories (34 questions) | Behavioral Health Disorders | Adult | Provider or Patient (typically) |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Kid Screener (ages 6-8) | Screens for 23 behavioral health categories (45 questions) | Behavioral Health Disorders | Children & Adolescents | Provider or Patient |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Kid Screener (ages 9-12) | Screens for 23 behavioral health categories (45 questions) | Behavioral Health Disorders | Children & Adolescents | Provider or Patient |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Kid Screener (ages 13-17) | Screens for 23 behavioral health categories (45 questions) | Behavioral Health Disorders | Children & Adolescents | Provider or Patient |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Senior Screener | Screens for 12 behavioral health categories prevalent in Seniors, including hoarding (21 questions) | Behavioral Health Disorders | Adult | Provider or Patient (typically) |
| Mood Disorder Screener | Determines if a patient may have a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders (9 questions) | Mood Disorders | Adult | Provider or Patient (typically) |
| PHQ-9 (Patient Health Questionnaire-9) | 9 questions used to measure the level of depression symptoms | Depression | Adult | Provider or Patient (typically) |
| PHQ-9 + GAD-7 (Patient Health Questionnaire-9 and Generalized Anxiety Disorder-7) | Combination of the PHQ-9 and GAD-7 in one tool | Depression and Anxiety | Adult | Provider or Patient (typically) |
| SUD (Substance Use Disorder) Screener | Determines if a patient may have an alcohol or drug use disorder (2 questions) | SUD | Adult | Provider or Patient |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
| ADHD Interview | An additional module that can be added to the M.I.N.I. Interview to cover ADHD | ADHD | Adult | Provider or Patient (typically) |
| Anxiety Disorder Interview | Determines if a patient meets criteria for an anxiety disorder, including Panic, Agorophobia, Social Anxiety, OCD, PTSD, GAD | Anxiety | Adult | Provider or Patient (typically) |
| Borderline Personality Disorder Interview | An additional module that can be added to the M.I.N.I. Interview to cover Borderline Personality Disorder | Borderline Peronality Disorder | Adult | Provider or Patient (typically) |
| C-SSRS (Columbia Suicide Severity Rating Scale) with SAFE-T | Suicidality interview that rates the suicidality risk level and offers the provider the appropriate actions to take based on the risk level | Suicidality | Adult | Provider |
| CY-BOCS (Children's Yale-Brown Obsessive Compulsive Scale) | OCD structured interview | OCD | Children & Adolescents | Provider |
| Eating Disorder Interview | Determines if a patient meets criteria for an eating disorder, including Anorexia, Bulimia, Binge Eating | Eating Disorders | Adult | Provider (typically) or Patient |
| Hoarding Interview | An additional module that can be added to the M.I.N.I. Interview to cover Hoarding | Hoarding | Adult | Provider or Patient (typically) |
| Kid Anxiety Disorder Interview | Interview that determines if a patient meets criteria for an anxiety disorder, including Panic, Agorophobia, Social Anxiety, OCD, PTSD, GAD | Anxiety | Children & Adolescents | Provider (typically) or Patient |
| Kid Conduct Interview | Interview that determines if a patient meets criteria for a conduct disorder | Conduct Disorder | Children & Adolescents | Provider (typically) or Patient |
| Kid Eating Disorder Interview | Interview that determines if a patient meets criteria for an eating disorder, including Anorexia, Bulimia, Binge Eating | Eating Disorders | Children & Adolescents | Provider (typically) or Patient |
| Kid Mood Disorder Interview | Interview that determines if a patient meets criteria for a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders | Mood Disorders | Children & Adolescents | Provider (typically) or Patient |
| Kid ODD Interview | Interview that determines if a patient meets criteria for an oppositional defiance disorder | Oppositional Defiance Disorder | Children & Adolescents | Provider (typically) or Patient |
| Kid SUD Interview | Interview that determines if a patient meets criteria for a substance use disorder | SUD | Children & Adolescents | Provider (typically) or Patient |
| Kid Tic Interview | Interview that determines if a patient meets criteria for a tic disorder | Tic Disorder | Children & Adolescents | Provider (typically) or Patient |
| Major Depressive Subtypes Interview | 5 additional modules that can be added on to a M.I.N.I. Interview for a deep analysis of Depression; example outcome: Major Depressive Episode with Anxious Distress | Depression | Adult | Provider or Patient (typically) |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Adult Interview | Rates 17 behavioral health categories, taking approximately 20 to 30 minutes to complete | Behavioral Health Disorders | Adult | Provider (typically) or Patient (will need to be edited by the provider to score all categories) |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Kid Interview | Rates 23 behavioral health categories, taking approximately 20 to 30 minutes to complete | Behavioral Health Disorders | Children & Adolescents | Provider (typically) or Patient (will need to be edited by the provider to score all categories) |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Kid Parent Interview | Rates 23 behavioral health categories, taking approximately 20 to 30 minutes to complete; question grammar is changed for a parent to answer the questions | Behavioral Health Disorders | Adult | Provider (typically) or Patient (will need to be edited by the provider to score all categories) |
| M.I.N.I. (Mini International Neuropsychiatric Interview) for Psychotic Disorders Interview | Along with the standard 17 categories in a M.I.N.I., this interview determines if a patient meets criteria for severe psychotic disorders such as Schizophrenia | Behavioral Health Disorders | Adult | Provider |
| M.I.N.I. (Mini International Neuropsychiatric Interview) Senior Interview | Rates disorders that are prevalent in Seniors, including hoarding | Behavioral Health Disorders | Adult | Provider (typically) or Patient |
| Mood Disorder Interview | Determines if a patient meets critera for a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders | Mood Disorders | Adult | Provider (typically) or Patient |
| SUD (Substance Use Disorder) Interview | Determines if a patient meets criteria for a substance use disorder | SUD | Adult | Provider (typically) or Patient |
| Y-BOCS I (Yale-Brown Obsessive Compulsive Scale) | OCD structured interview | OCD | Adult | Provider |
| Y-BOCS II (Yale-Brown Obsessive Compulsive Scale) | Updated OCD structured interview | OCD | Adult | Provider |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
| Adult Agoraphobia Monitor | All Monitors include symptomology questions for the represented disorder, which can be charted for measuring success of treatment plans. | Agoraphobia | Adult | Provider or Patient (typically) |
| Adult Alcohol Use Monitor | Alcohol Use | Adult | Provider or Patient (typically) | |
| Adult Anorexia Monitor | Anorexia | Adult | Provider or Patient (typically) | |
| Adult Antisocial Personality Disorder Monitor | Antisocial Personality Disorder | Adult | Provider or Patient (typically) | |
| Adult Binge Eating Monitor | Binge Eating | Adult | Provider or Patient (typically) | |
| Adult Bulimia Monitor | Bulimia | Adult | Provider or Patient (typically) | |
| Adult Depression Monitor | Depression | Adult | Provider or Patient (typically) | |
| Adult GAD Monitor | GAD | Adult | Provider or Patient (typically) | |
| Adult Manic Disorder Monitor | Manic Disorder | Adult | Provider or Patient (typically) | |
| Adult OCD Monitor | OCD | Adult | Provider or Patient (typically) | |
| Adult Panic Disorder Monitor | Panic Disorder | Adult | Provider or Patient (typically) | |
| Adult Psychosis Monitor | Psychosis | Adult | Provider or Patient (typically) | |
| Adult PTSD Monitor | PTSD | Adult | Provider or Patient (typically) | |
| Adult Social Anxiety Monitor | Social Anxiety | Adult | Provider or Patient (typically) | |
| Adult SUD Monitor | SUD | Adult | Provider or Patient (typically) | |
| Adult Suicidality Monitor | Suicidality | Adult | Provider or Patient (typically) | |
| BARC-10 (Brief Assessment of Recovery Capital) | Measures the "recovery capital" to predict the likelihood of a patient staying in remission | SUD | Adult | Patient |
| GAD-7 (Generalized Anxiety Disorder-7) | 7 questions used to measure the level of Generalized Anxiety Disorder symptoms | Anxiety | Adult | Provider or Patient (typically) |
| Kid ADHD Monitor | ADHD | Children & Adolescents | Provider or Patient (typically) | |
| Kid Adjustment Disorder | Adjustment | Children & Adolescents | Provider or Patient (typically) | |
| Kid Agoraphobia Monitor | Agoraphobia | Children & Adolescents | Provider or Patient (typically) | |
| Kid Alcohol Use Monitor | Alcohol Use | Children & Adolescents | Provider or Patient (typically) | |
| Kid Anorexia Monitor | Anorexia | Children & Adolescents | Provider or Patient (typically) | |
| Kid Antisocial Personality Disorder Monitor | Antisocial Personality Disorder | Children & Adolescents | Provider or Patient (typically) | |
| Kid Binge Eating Monitor | Binge Eating | Children & Adolescents | Provider or Patient (typically) | |
| Kid Bulimia Monitor | Bulimia | Children & Adolescents | Provider or Patient (typically) | |
| Kid Conduct Disorder Monitor | Conduct | Children & Adolescents | Provider or Patient (typically) | |
| Kid Depression Monitor | Depression | Children & Adolescents | Provider or Patient (typically) | |
| Kid GAD Monitor | GAD | Children & Adolescents | Provider or Patient (typically) | |
| Kid Manic Disorder Monitor | Manic Disorder | Children & Adolescents | Provider or Patient (typically) | |
| Kid OCD Monitor | OCD | Children & Adolescents | Provider or Patient (typically) | |
| Kid Oppositional Defiant Disorder Monitor | Oppositional Defiant | Children & Adolescents | Provider or Patient (typically) | |
| Kid Panic Disorder Monitor | Panic Disorder | Children & Adolescents | Provider or Patient (typically) | |
| Kid Psychosis Monitor | Psychosis | Children & Adolescents | Provider or Patient (typically) | |
| Kid PTSD Monitor | PTSD | Children & Adolescents | Provider or Patient (typically) | |
| Kid Separation Anxiety Monitor | Separation Anxiety | Children & Adolescents | Provider or Patient (typically) | |
| Kid Social Anxiety Monitor | Social Anxiety | Children & Adolescents | Provider or Patient (typically) | |
| Kid Specific Phobia Monitor | Specific Phobia | Children & Adolescents | Provider or Patient (typically) | |
| Kid SUD Monitor | SUD | Children & Adolescents | Provider or Patient (typically) | |
| Kid Suicidality Monitor (ages 6-8) | Suicidality | Children & Adolescents | Provider or Patient (typically) | |
| Kid Suicidality Monitor (ages 9-12) | Suicidality | Children & Adolescents | Provider or Patient (typically) | |
| Kid Suicidality Monitor (ages 13-19) | Suicidality | Children & Adolescents | Provider or Patient (typically) | |
| Kid Tic Disorders Monitor | Tic Disorders | Children & Adolescents | Provider or Patient (typically) | |
| PHQ-9 (Patient Health Questionnaire-9) | 9 questions used to measure the level of depression symptoms | Depression | Adult | Provider or Patient (typically) |
| PHQ-9 + GAD-7 (Patient Health Questionnaore-9 and Generalized Anxiety Disorder-7) | Combination of the PHQ-9 and GAD-7 in one tool | Depression and Anxiety | Adult | Provider or Patient (typically) |
| PSS (Perceived Stress Assessment) | Rates a patient's stress level as low, moderate, or high (10 questions) | Stress | Adult | Provider or Patient |
| PSQI (Pittsburgh Sleep Index) | Assesses sleep quality over a 1-month time interval; measures 7 components of sleep (26 questions) | Sleep | Adult | Provider or Patient |
| S-DISS (Screener for Dissociation) | Asks if the patient has a foggy mind, is detached, feels outside of his/her body (1 question) | Dissociation | Adult | Provider or Patient (typically) |
| SDS (Sheehan Disability Scale) | Functional assessment that asks about how a patient is functioning at home, work or school, and number of productive days (5 questions) | Functionality | Adult | Provider or Patient (typically) |
| SIS (Sheehan Irritability Scale) | Allows the patient to rate the severity level of 7 different irritability manifestations and the number of days his/her irritability has impaired daily life (8 questions) | Irritability | Adult | Patient |
| SPS (Sheehan Panic Scale) | Allows the patient to rate the severity level of 35 different manifestations of panic which helps the provider understand how the panic is affecting the patient's daily life and what his/her symptoms are (35 questions) | Panic Disorder | Adult | Patient |
| S-STS (Sheehan Suicidality Tracking Scale) | Deep dive into a patient's sucidality history; delivers a score; can be used in an emergency setting to route a patient to appropriate treatment | Suicidality | Adult | Provider (typically) or Patient |
| S-STS (Sheehan Suicidality Tracking Scale) Short Version | Same as the S-STS except this version does not include questions about appointments missed | Suicidality | Adult | Provider (typically) or Patient |
| Therapeutic Alliance | Measures the relationship between the patient and provider, which is an important factor for successful behavioral health treatment | Alliance to provider | Adult | Patient |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
| ACEs (Adverse Childhood Experiences) | Delivers a score based on childhood circumstances such as childhood abuse, environment, and economic stability that can affect a wide range of health, functioning and quality of life outcomes and risks in adults and can impact the likelihood of behavioral health disorders | Childhood experiences | Adult | Provider or Patient (typically) |
| AQ-10 (Autism Spectrum Quotient) | A screener that provides a quick referral guide for adults with suspected autism who do not have a learning disability (10 questions) | Autism | Adult | Patient |
| AQ-50 (Autism Spectrum Quotient) | An interview that may indicated a patient might be autistic depending on how it is answered (50 questions) | Autism | Adult | Patient |
| BARC-10 (Brief Assessment of Recovery Capital) | Measures the "recovery capital" to predict the likelihood of a SUD patient staying in remission (10 questions) | SUD | Adult | Patient |
| HRSN (Health-Related Social Needs) Screening Tool | Asks questions about health-related social needs to determine effect on total healthcare costs and better outcomes; 5 core domains include: housing instability, food insecurity, transportation problems, utility needs, and interpersonal safety; 8 supplemental domains include: financial strain, employment, family and community support, education, physical activity, substance use, mental health, and disabilities | SUD | Adult | Patient or Caregiver (typically) |
| PEARLS - Teen (Caregiver) | Screener for childhood experiences that affect a wide range of health, functioning and quality of life outcomes and risks | Childhood experiences | Teen | Caregiver |
| PEARLS - Teen (Self Report) | Screener for childhood experiences that affect a wide range of health, functioning and quality of life outcomes and risks | Childhood experiences | Teen | Patient |
| PSS (Perceived Stress Assessment) | Rates a patient's stress level as low, moderate, or high (10 questions) | Stress | Adult | Provider or Patient |
| SDOH (Social Determinants of Health) | Asks questions about enviornmental conditions that affect a wide range of health, functioning and quality of life outcomes and risks; areas include: economic stability, education access and quality, healthcare access and quality, neighborhood enviornment, social and community context (50 questions) | Environmental conditions | Adult | Provider or Patient |
| Urge to Use Scale | Measures the "urge to use" aspect of recovery, asking questions in the timeframe of the past week (6 questions) | SUD | Adult | Provider or Patient (typically) |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
ToolAnxiety Disorder Screener |
DescriptionDetermines if a patient may have an anxiety disorder, including Panic, Agorophobia, Social Anxiety, OCD, PTSD, GAD (9 questions) |
DisorderAnxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolBDDQ (Body Dysmorphic Disorder Questionnaire) |
DescriptionDetermines if a person may have Body Dysmorphic Disorder |
DisorderBDD |
Patient AgeAdult |
How AdministeredPatient |
ToolC-FOCI (Children's Obsessive Compulsive Inventory) |
DescriptionOCD symptoms experienced in the past month (24 questions) |
DisorderOCD |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient |
ToolC-SSRS (Columbia Suicide Severity Rating Scale) |
DescriptionSuicidality screener (5 questions) |
DisorderSuicidality |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolEating Disorder Screener |
DescriptionDetermines if a patient may have an eating disorder, including Anorexia, Bulimia, Binge Eating (4 questions) |
DisorderEating Disorders |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolFOCI (Florida Obsessive Compulsive Inventory) |
DescriptionOCD symptoms experienced in the past month (25 questions) |
DisorderOCD |
Patient AgeAdult |
How AdministeredProvider or Patient |
ToolGAD-7 (Generalized Anxiety Disorder-7) |
Description7 questions used to measure the level of Generalized Anxiety Disorder symptoms |
DisorderAnxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolKid Mood Disorder Screener Ages 13-17 |
DescriptionDetermines if a patient may have a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders (9 questions) |
DisorderMood Disorders |
Patient AgeAdolescents |
How AdministeredProvider or Patient (typically) |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Adult Screener |
DescriptionScreens for 17 behavioral health categories (34 questions) |
DisorderBehavioral Health Disorders |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Kid Screener (ages 6-8) |
DescriptionScreens for 23 behavioral health categories (45 questions) |
DisorderBehavioral Health Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Kid Screener (ages 9-12) |
DescriptionScreens for 23 behavioral health categories (45 questions) |
DisorderBehavioral Health Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Kid Screener (ages 13-17) |
DescriptionScreens for 23 behavioral health categories (45 questions) |
DisorderBehavioral Health Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Senior Screener |
DescriptionScreens for 12 behavioral health categories prevalent in Seniors, including hoarding (21 questions) |
DisorderBehavioral Health Disorders |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolMood Disorder Screener |
DescriptionDetermines if a patient may have a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders (9 questions) |
DisorderMood Disorders |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolPHQ-9 (Patient Health Questionnaire-9) |
Description9 questions used to measure the level of depression symptoms |
DisorderDepression |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolPHQ-9 + GAD-7 (Patient Health Questionnaire-9 and Generalized Anxiety Disorder-7) |
DescriptionCombination of the PHQ-9 and GAD-7 in one tool |
DisorderDepression and Anxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolSUD (Substance Use Disorder) Screener |
DescriptionDetermines if a patient may have an alcohol or drug use disorder (2 questions) |
DisorderSUD |
Patient AgeAdult |
How AdministeredProvider or Patient |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
ToolADHD Interview |
DescriptionAn additional module that can be added to the M.I.N.I. Interview to cover ADHD |
DisorderADHD |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAnxiety Disorder Interview |
DescriptionDetermines if a patient meets criteria for an anxiety disorder, including Panic, Agorophobia, Social Anxiety, OCD, PTSD, GAD |
DisorderAnxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolBorderline Personality Disorder Interview |
DescriptionAn additional module that can be added to the M.I.N.I. Interview to cover Borderline Personality Disorder |
DisorderBorderline Peronality Disorder |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolC-SSRS (Columbia Suicide Severity Rating Scale) with SAFE-T |
DescriptionSuicidality interview that rates the suicidality risk level and offers the provider the appropriate actions to take based on the risk level |
DisorderSuicidality |
Patient AgeAdult |
How AdministeredProvider |
ToolCY-BOCS (Children's Yale-Brown Obsessive Compulsive Scale) |
DescriptionOCD structured interview |
DisorderOCD |
Patient AgeChildren & Adolescents |
How AdministeredProvider |
ToolEating Disorder Interview |
DescriptionDetermines if a patient meets criteria for an eating disorder, including Anorexia, Bulimia, Binge Eating |
DisorderEating Disorders |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient |
ToolHoarding Interview |
DescriptionAn additional module that can be added to the M.I.N.I. Interview to cover Hoarding |
DisorderHoarding |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolKid Anxiety Disorder Interview |
DescriptionInterview that determines if a patient meets criteria for an anxiety disorder, including Panic, Agorophobia, Social Anxiety, OCD, PTSD, GAD |
DisorderAnxiety |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolKid Conduct Interview |
DescriptionInterview that determines if a patient meets criteria for a conduct disorder |
DisorderConduct Disorder |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolKid Eating Disorder Interview |
DescriptionInterview that determines if a patient meets criteria for an eating disorder, including Anorexia, Bulimia, Binge Eating |
DisorderEating Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolKid Mood Disorder Interview |
DescriptionInterview that determines if a patient meets criteria for a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders |
DisorderMood Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolKid ODD Interview |
DescriptionInterview that determines if a patient meets criteria for an oppositional defiance disorder |
DisorderOppositional Defiance Disorder |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolKid SUD Interview |
DescriptionInterview that determines if a patient meets criteria for a substance use disorder |
DisorderSUD |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolKid Tic Interview |
DescriptionInterview that determines if a patient meets criteria for a tic disorder |
DisorderTic Disorder |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient |
ToolMajor Depressive Subtypes Interview |
Description5 additional modules that can be added on to a M.I.N.I. Interview for a deep analysis of Depression; example outcome: Major Depressive Episode with Anxious Distress |
DisorderDepression |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Adult Interview |
DescriptionRates 17 behavioral health categories, taking approximately 20 to 30 minutes to complete |
DisorderBehavioral Health Disorders |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient (will need to be edited by the provider to score all categories) |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Kid Interview |
DescriptionRates 23 behavioral health categories, taking approximately 20 to 30 minutes to complete |
DisorderBehavioral Health Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider (typically) or Patient (will need to be edited by the provider to score all categories) |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Kid Parent Interview |
DescriptionRates 23 behavioral health categories, taking approximately 20 to 30 minutes to complete; question grammar is changed for a parent to answer the questions |
DisorderBehavioral Health Disorders |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient (will need to be edited by the provider to score all categories) |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) for Psychotic Disorders Interview |
DescriptionAlong with the standard 17 categories in a M.I.N.I., this interview determines if a patient meets criteria for severe psychotic disorders such as Schizophrenia |
DisorderBehavioral Health Disorders |
Patient AgeAdult |
How AdministeredProvider |
ToolM.I.N.I. (Mini International Neuropsychiatric Interview) Senior Interview |
DescriptionRates disorders that are prevalent in Seniors, including hoarding |
DisorderBehavioral Health Disorders |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient |
ToolMood Disorder Interview |
DescriptionDetermines if a patient meets critera for a mood disorder, including Depression, Mania, Psychosis, Bipolar Disorders |
DisorderMood Disorders |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient |
ToolSUD (Substance Use Disorder) Interview |
DescriptionDetermines if a patient meets criteria for a substance use disorder |
DisorderSUD |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient |
ToolY-BOCS I (Yale-Brown Obsessive Compulsive Scale) |
DescriptionOCD structured interview |
DisorderOCD |
Patient AgeAdult |
How AdministeredProvider |
ToolY-BOCS II (Yale-Brown Obsessive Compulsive Scale) |
DescriptionUpdated OCD structured interview |
DisorderOCD |
Patient AgeAdult |
How AdministeredProvider |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
ToolAdult Agoraphobia Monitor |
DescriptionAll Monitors include symptomology questions for the represented disorder, which can be charted for measuring success of treatment plans. |
DisorderAgoraphobia |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Alcohol Use Monitor |
Description |
DisorderAlcohol Use |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Anorexia Monitor |
Description |
DisorderAnorexia |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Antisocial Personality Disorder Monitor |
Description |
DisorderAntisocial Personality Disorder |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Binge Eating Monitor |
Description |
DisorderBinge Eating |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Bulimia Monitor |
Description |
DisorderBulimia |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Depression Monitor |
Description |
DisorderDepression |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult GAD Monitor |
Description |
DisorderGAD |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Manic Disorder Monitor |
Description |
DisorderManic Disorder |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult OCD Monitor |
Description |
DisorderOCD |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Panic Disorder Monitor |
Description |
DisorderPanic Disorder |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Psychosis Monitor |
Description |
DisorderPsychosis |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult PTSD Monitor |
Description |
DisorderPTSD |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Social Anxiety Monitor |
Description |
DisorderSocial Anxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult SUD Monitor |
Description |
DisorderSUD |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAdult Suicidality Monitor |
Description |
DisorderSuicidality |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolBARC-10 (Brief Assessment of Recovery Capital) |
DescriptionMeasures the "recovery capital" to predict the likelihood of a patient staying in remission |
DisorderSUD |
Patient AgeAdult |
How AdministeredPatient |
ToolGAD-7 (Generalized Anxiety Disorder-7) |
Description7 questions used to measure the level of Generalized Anxiety Disorder symptoms |
DisorderAnxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolKid ADHD Monitor |
Description |
DisorderADHD |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Adjustment Disorder |
Description |
DisorderAdjustment |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Agoraphobia Monitor |
Description |
DisorderAgoraphobia |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Alcohol Use Monitor |
Description |
DisorderAlcohol Use |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Anorexia Monitor |
Description |
DisorderAnorexia |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Antisocial Personality Disorder Monitor |
Description |
DisorderAntisocial Personality Disorder |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Binge Eating Monitor |
Description |
DisorderBinge Eating |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Bulimia Monitor |
Description |
DisorderBulimia |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Conduct Disorder Monitor |
Description |
DisorderConduct |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Depression Monitor |
Description |
DisorderDepression |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid GAD Monitor |
Description |
DisorderGAD |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Manic Disorder Monitor |
Description |
DisorderManic Disorder |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid OCD Monitor |
Description |
DisorderOCD |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Oppositional Defiant Disorder Monitor |
Description |
DisorderOppositional Defiant |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Panic Disorder Monitor |
Description |
DisorderPanic Disorder |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Psychosis Monitor |
Description |
DisorderPsychosis |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid PTSD Monitor |
Description |
DisorderPTSD |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Separation Anxiety Monitor |
Description |
DisorderSeparation Anxiety |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Social Anxiety Monitor |
Description |
DisorderSocial Anxiety |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Specific Phobia Monitor |
Description |
DisorderSpecific Phobia |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid SUD Monitor |
Description |
DisorderSUD |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Suicidality Monitor (ages 6-8) |
Description |
DisorderSuicidality |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Suicidality Monitor (ages 9-12) |
Description |
DisorderSuicidality |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Suicidality Monitor (ages 13-19) |
Description |
DisorderSuicidality |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolKid Tic Disorders Monitor |
Description |
DisorderTic Disorders |
Patient AgeChildren & Adolescents |
How AdministeredProvider or Patient (typically) |
ToolPHQ-9 (Patient Health Questionnaire-9) |
Description9 questions used to measure the level of depression symptoms |
DisorderDepression |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolPHQ-9 + GAD-7 (Patient Health Questionnaore-9 and Generalized Anxiety Disorder-7) |
DescriptionCombination of the PHQ-9 and GAD-7 in one tool |
DisorderDepression and Anxiety |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolPSS (Perceived Stress Assessment) |
DescriptionRates a patient's stress level as low, moderate, or high (10 questions) |
DisorderStress |
Patient AgeAdult |
How AdministeredProvider or Patient |
ToolPSQI (Pittsburgh Sleep Index) |
DescriptionAssesses sleep quality over a 1-month time interval; measures 7 components of sleep (26 questions) |
DisorderSleep |
Patient AgeAdult |
How AdministeredProvider or Patient |
ToolS-DISS (Screener for Dissociation) |
DescriptionAsks if the patient has a foggy mind, is detached, feels outside of his/her body (1 question) |
DisorderDissociation |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolSDS (Sheehan Disability Scale) |
DescriptionFunctional assessment that asks about how a patient is functioning at home, work or school, and number of productive days (5 questions) |
DisorderFunctionality |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolSIS (Sheehan Irritability Scale) |
DescriptionAllows the patient to rate the severity level of 7 different irritability manifestations and the number of days his/her irritability has impaired daily life (8 questions) |
DisorderIrritability |
Patient AgeAdult |
How AdministeredPatient |
ToolSPS (Sheehan Panic Scale) |
DescriptionAllows the patient to rate the severity level of 35 different manifestations of panic which helps the provider understand how the panic is affecting the patient's daily life and what his/her symptoms are (35 questions) |
DisorderPanic Disorder |
Patient AgeAdult |
How AdministeredPatient |
ToolS-STS (Sheehan Suicidality Tracking Scale) |
DescriptionDeep dive into a patient's sucidality history; delivers a score; can be used in an emergency setting to route a patient to appropriate treatment |
DisorderSuicidality |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient |
ToolS-STS (Sheehan Suicidality Tracking Scale) Short Version |
DescriptionSame as the S-STS except this version does not include questions about appointments missed |
DisorderSuicidality |
Patient AgeAdult |
How AdministeredProvider (typically) or Patient |
ToolTherapeutic Alliance |
DescriptionMeasures the relationship between the patient and provider, which is an important factor for successful behavioral health treatment |
DisorderAlliance to provider |
Patient AgeAdult |
How AdministeredPatient |
| Tool | Description | Disorder | Patient Age | How Administered |
|---|---|---|---|---|
ToolACEs (Adverse Childhood Experiences) |
DescriptionDelivers a score based on childhood circumstances such as childhood abuse, environment, and economic stability that can affect a wide range of health, functioning and quality of life outcomes and risks in adults and can impact the likelihood of behavioral health disorders |
DisorderChildhood experiences |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
ToolAQ-10 (Autism Spectrum Quotient) |
DescriptionA screener that provides a quick referral guide for adults with suspected autism who do not have a learning disability (10 questions) |
DisorderAutism |
Patient AgeAdult |
How AdministeredPatient |
ToolAQ-50 (Autism Spectrum Quotient) |
DescriptionAn interview that may indicated a patient might be autistic depending on how it is answered (50 questions) |
DisorderAutism |
Patient AgeAdult |
How AdministeredPatient |
ToolBARC-10 (Brief Assessment of Recovery Capital) |
DescriptionMeasures the "recovery capital" to predict the likelihood of a SUD patient staying in remission (10 questions) |
DisorderSUD |
Patient AgeAdult |
How AdministeredPatient |
ToolHRSN (Health-Related Social Needs) Screening Tool |
DescriptionAsks questions about health-related social needs to determine effect on total healthcare costs and better outcomes; 5 core domains include: housing instability, food insecurity, transportation problems, utility needs, and interpersonal safety; 8 supplemental domains include: financial strain, employment, family and community support, education, physical activity, substance use, mental health, and disabilities |
DisorderSUD |
Patient AgeAdult |
How AdministeredPatient or Caregiver (typically) |
ToolPEARLS - Teen (Caregiver) |
DescriptionScreener for childhood experiences that affect a wide range of health, functioning and quality of life outcomes and risks |
DisorderChildhood experiences |
Patient AgeTeen |
How AdministeredCaregiver |
ToolPEARLS - Teen (Self Report) |
DescriptionScreener for childhood experiences that affect a wide range of health, functioning and quality of life outcomes and risks |
DisorderChildhood experiences |
Patient AgeTeen |
How AdministeredPatient |
ToolPSS (Perceived Stress Assessment) |
DescriptionRates a patient's stress level as low, moderate, or high (10 questions) |
DisorderStress |
Patient AgeAdult |
How AdministeredProvider or Patient |
ToolSDOH (Social Determinants of Health) |
DescriptionAsks questions about enviornmental conditions that affect a wide range of health, functioning and quality of life outcomes and risks; areas include: economic stability, education access and quality, healthcare access and quality, neighborhood enviornment, social and community context (50 questions) |
DisorderEnvironmental conditions |
Patient AgeAdult |
How AdministeredProvider or Patient |
ToolUrge to Use Scale |
DescriptionMeasures the "urge to use" aspect of recovery, asking questions in the timeframe of the past week (6 questions) |
DisorderSUD |
Patient AgeAdult |
How AdministeredProvider or Patient (typically) |
Proven Tools
Evidence-based Measurements Backed by Years of Research
The Proem platform is built using digital versions of tools that are exclusively licensed from the top psychiatric professionals in the behavioral health field. These tools include the Mini International Neuropsychiatric Interview (M.I.N.I.), Yale-Brown Obsessive Compulsive Scale (YBOCS), Body Dysmorphic Disorder Questionnaire (BDDQ), and more, and have been validated through research and used in clinical practice for many years worldwide.
Use Cases

Study: Partners in Caring for Anxious Youth, funded by Patient Center Outcomes Research Institute.
The study is a clinical trial comparing the effectiveness of evidence-based treatments for child and adolescent anxiety. The study compares the use of cognitive behavioral therapy (CBT) alone to CBT plus medication treatment to see which is more effective at achieving anxiety symptom remission and functional improvement at 6 and 12 months. Proem licensed digital assessments are being used to eliminate bias when selecting patients for the study.
Ann and Robert H. Lurie Children’s Hospital of Chicago

Study: The Australian Child Maltreatment Study.
This study uses data gathered from Proem licensed digital assessments to study 8,500 Australians who experienced child abuse and neglect in their childhood and adolescence to examine associations with a wide range of physical and mental health outcomes. The study is one of the most comprehensive studies on child maltreatment ever conducted and is intended to inform policy and practice reforms to reduce child abuse and neglect in Australia.
Queensland University of Technology

Study: The Childhood to Adolescence Transition Study.
This study uses the Proem licensed digital child assessment to gather data for a longitudinal study of children as they move into high school and through adolescence to understand the many influences on the health and emotional adjustment of children as they approach their teens. This is the first study in Melbourne to look at a population sample of children with this focus.
Murdoch Children’s Research Institute
Frequently Asked Questions
Proem digital tools can help researchers more accurately and efficiently recruit study participants by reducing bias and scoring errors. Proem digital tools can also help identify co-morbidities that may negatively affect trial outcomes if not initially identified.
Proem digital tools will decrease the amount of time needed to recruit trial candidates as the assessments are automatically scored and results are available within minutes of completion of the assessment.
One of the more commonly used Proem tools, the M.I.N.I., assesses the 17 most common adult disorders, and the assessment can be configured to use only the modules needed for a particular study. This saves the researcher time as multiple assessments are not necessary for candidate recruitment.
The cost ranges from $4 to $15 per screener or assessment depending on the tool and number purchased plus a small one-time implementation fee if using the Proem platform. Proem licenses the tools that it digitizes and distributes via its platform from world-renowned psychiatrists and psychologists who authored and maintain the tools. Therefore, there is a cost associated with the tools.
One mission. Two ways.
At Proem Behavioral Health, our mission is to ease the pain of mental illness by helping healthcare providers and clinical researchers help more people. We do that in two ways: through our comprehensive software platform and our exclusively licensed digital mental health assessments.
The Proem Platform
Experience workflow efficiencies, consistent processes and documented outcome measures with a digital behavioral health assessment platform.
Gold-standard Measurements
Just need access to the best mental health assessment tools available to help screen, diagnose and/or monitor patients or trial/study participants?